
SOLVED: What is the role of the thiosulfate in this experiment " Oxidation of Iodide with hydrogen peroxide " ?

SOLVED: The reaction between dilute sodium thiosulfate and hydrogen peroxide in the aqueous solution is irreversible and is second order in thiosulfate. The reaction of the peroxide in the dilute aqueous phase

Ch. 20 Applications of oxidation/reduction titrations 1.Auxiliary oxidizing and reducing reagents Fe(II), Fe(III) - Auxiliary reducing reagents A number. - ppt download

SOLVED: The reaction between sodium thiosulfate and hydrogen peroxide in dilute aqueous solution is irreversible and second order in thiosulfate. The reaction of sodium thiosulfate and hydrogen peroxide in the dilute aqueous

Time profile of the reaction of thiosulfate with hydrogen peroxide by... | Download Scientific Diagram

A 2.00 mL sample of an aqueous solution of hydrogen peroxide, H2O2(aq), is treated with an excess of Kl(aq). The liberated I2 requires 12.40 mL of 0.1025 M Na2S2O3 for its titration.
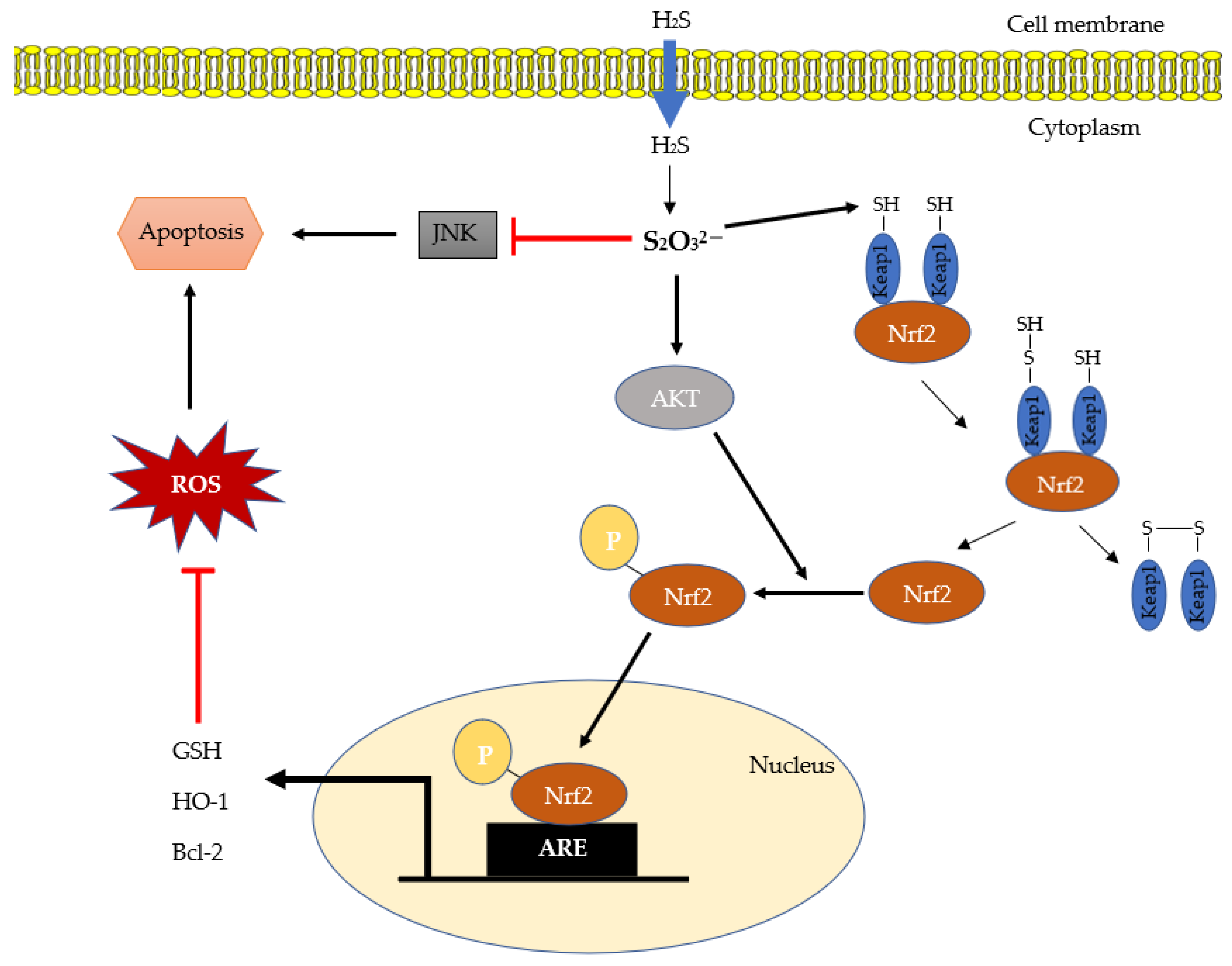
IJMS | Free Full-Text | Hydrogen Sulfide Metabolite, Sodium Thiosulfate: Clinical Applications and Underlying Molecular Mechanisms

SOLVED: Background: This oscillating clock examines the reaction between iodide, hydrogen ion, and hydrogen peroxide. The reaction order of hydrogen peroxide will be shown to be first order, and the rate constant (

Amazon.com: LabChem LC250604 Sodium Thiosulfate Solution, 0.1N (0.1M), 4 L Volume : Industrial & Scientific


















