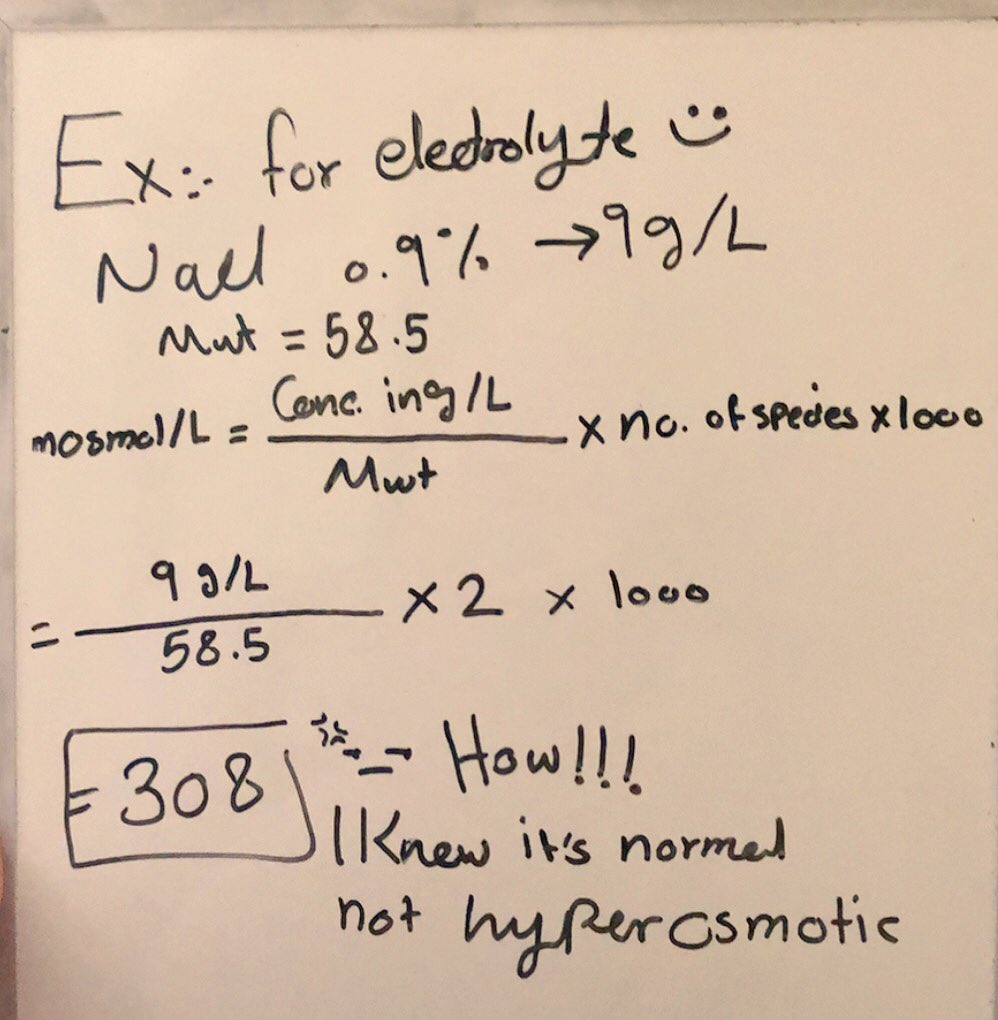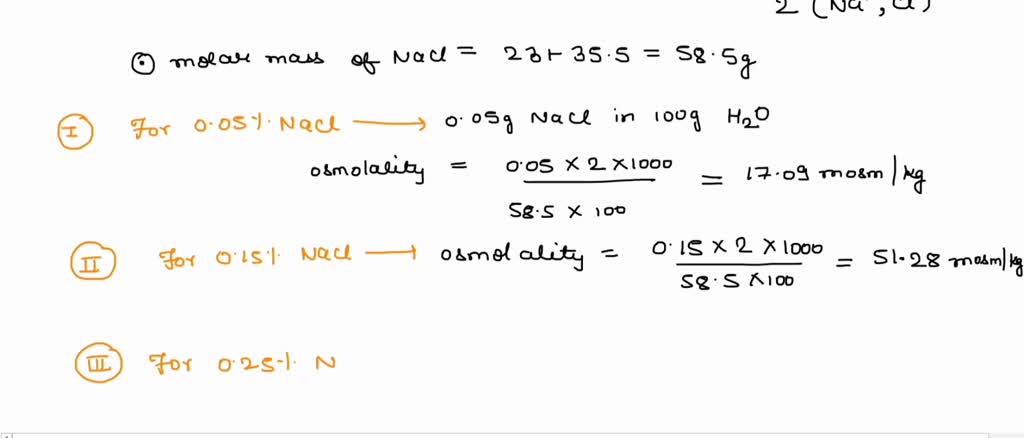
SOLVED: Calculate the osmolality for each of these six solutions 0.05, 0.15, 0.25, 0.6, 0.9, 1.2% NaCl. Which concentration(s) is/are closest to that of plasma (290 mOsm/kg)?

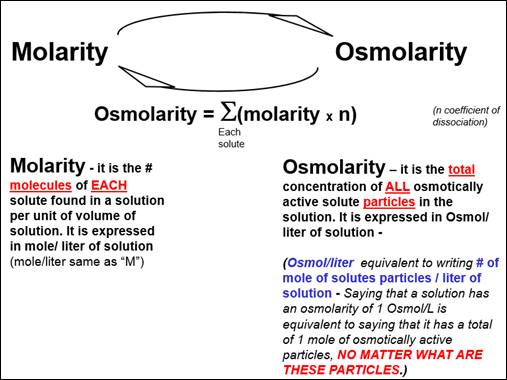
SOLVED: If a solution contains 100 mM glucose and 100 mM NaCl, what is the osmolarity of the solution? 400 mOsM 0.3 OsM 300 mM D. 200 mM 200 mOsM

What is the molal concentration of a 0.9% NaCl solution? What is the osmolar concentration? | Homework.Study.com
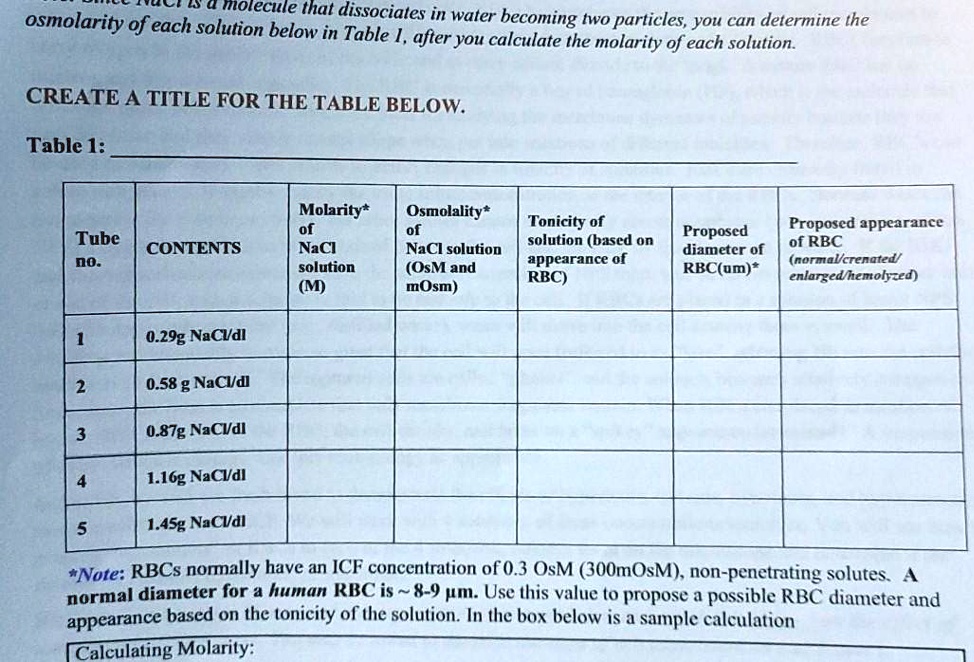
SOLVED: 0 4 molecule that osmolarity of each solution below dissociates in water becoming twO particles, you can determine the in Table I, after you calculate the molarity of each solution. CREATE

SOLVED: Calculatlng Osmolarity Prectke Wbrksheet Calculate the osmolarity of a 0.3 M NaCl solution: Calculate the osmolarity ofa 0.5 M glucose, 0.1 M NaCl solution Calculate the osmolarity ofa 0.2 M sucrose,