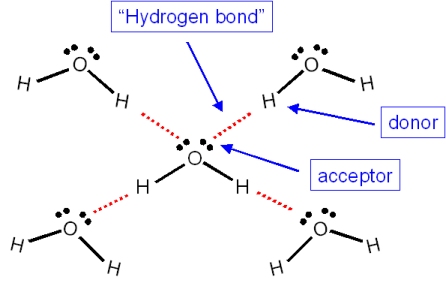
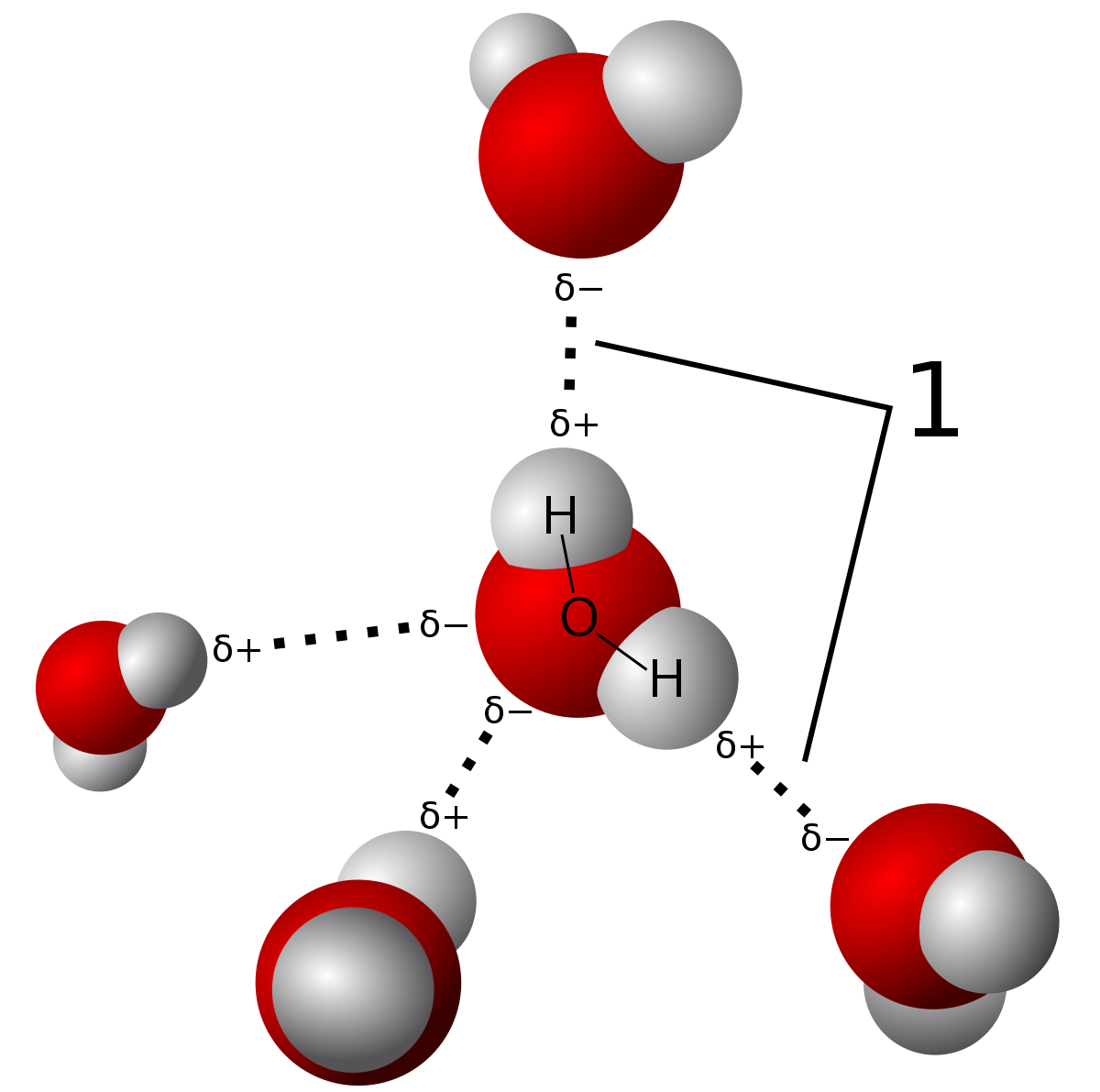
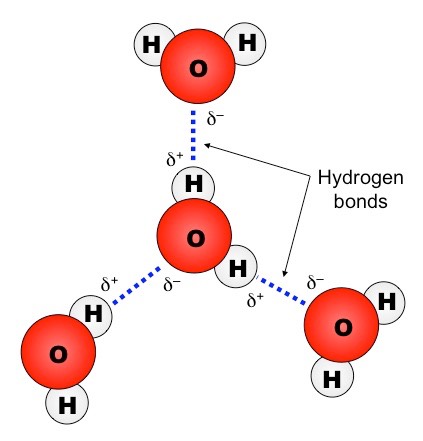
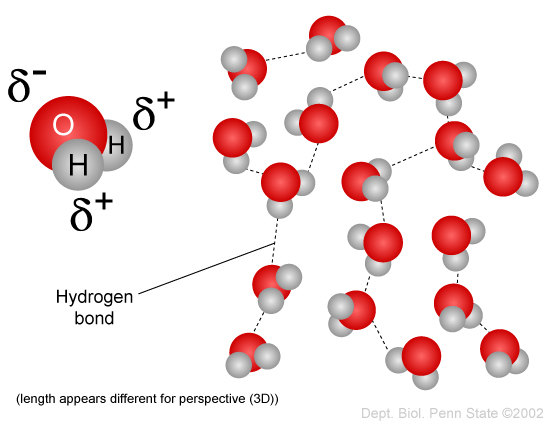
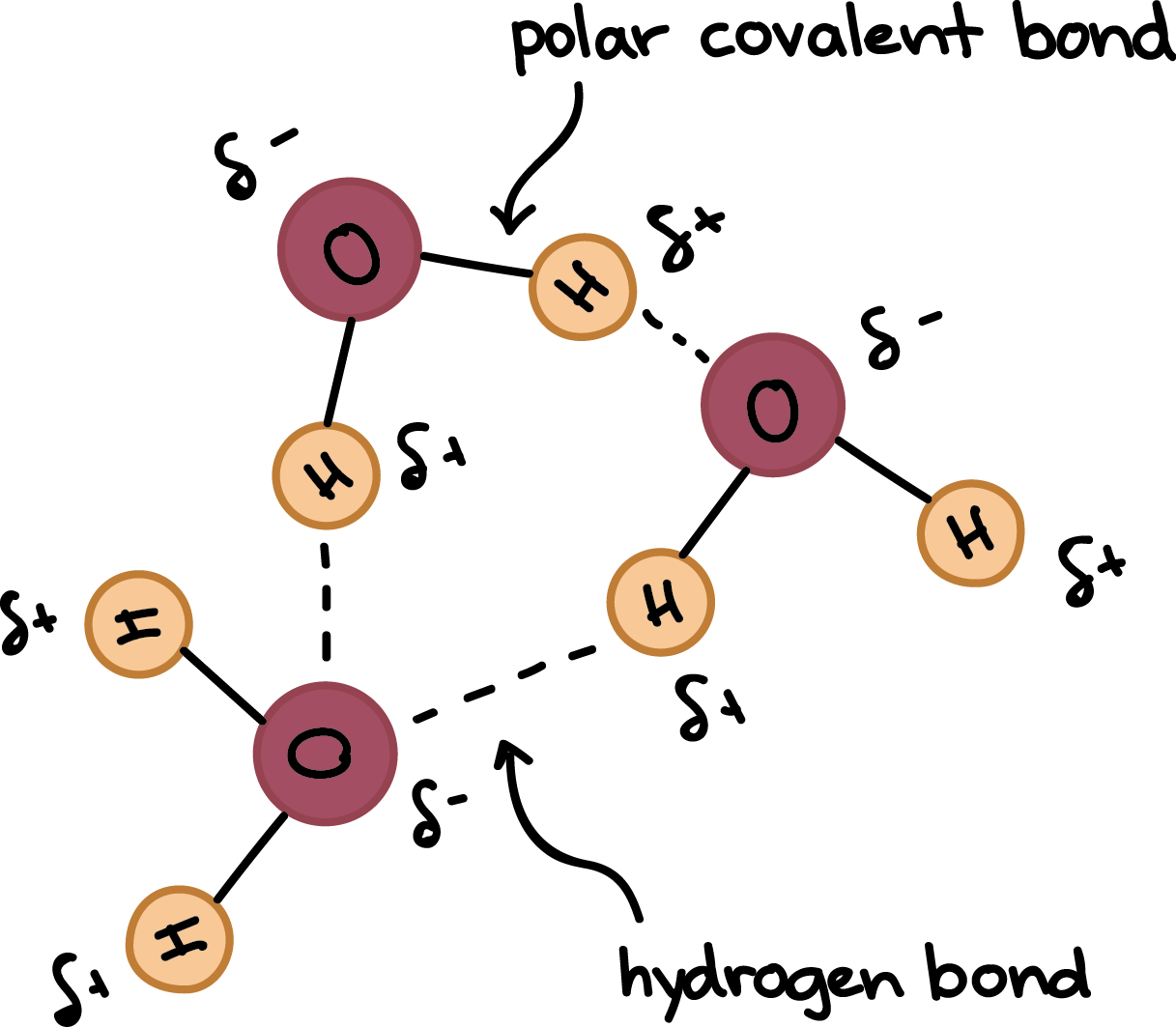On average, how many hydrogen bonds does any water molecule form with neighboring waters in liquid water? Draw a diagram of water H-bonding. | Homework.Study.com
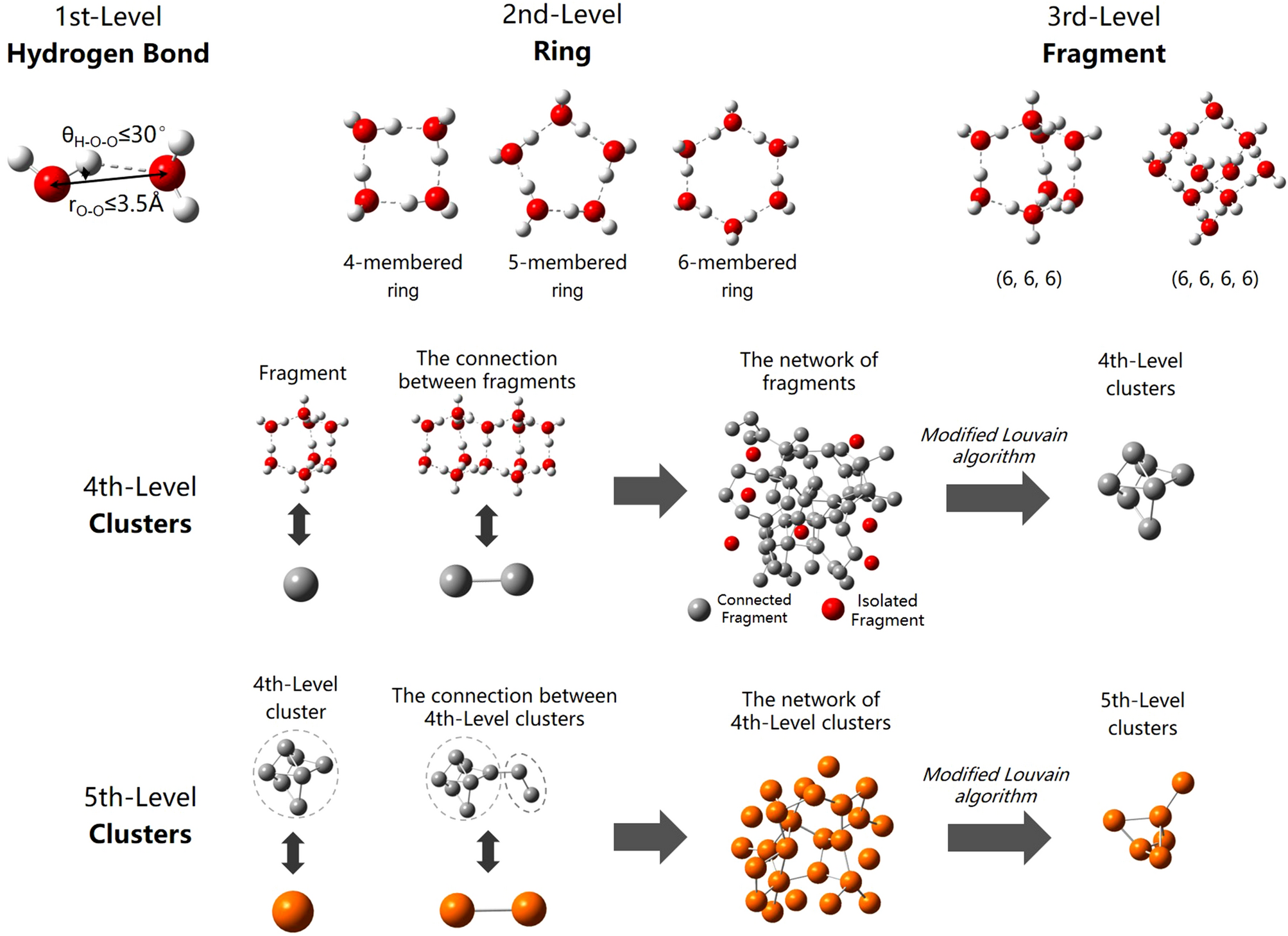
A hierarchical clustering method of hydrogen bond networks in liquid water undergoing shear flow | Scientific Reports

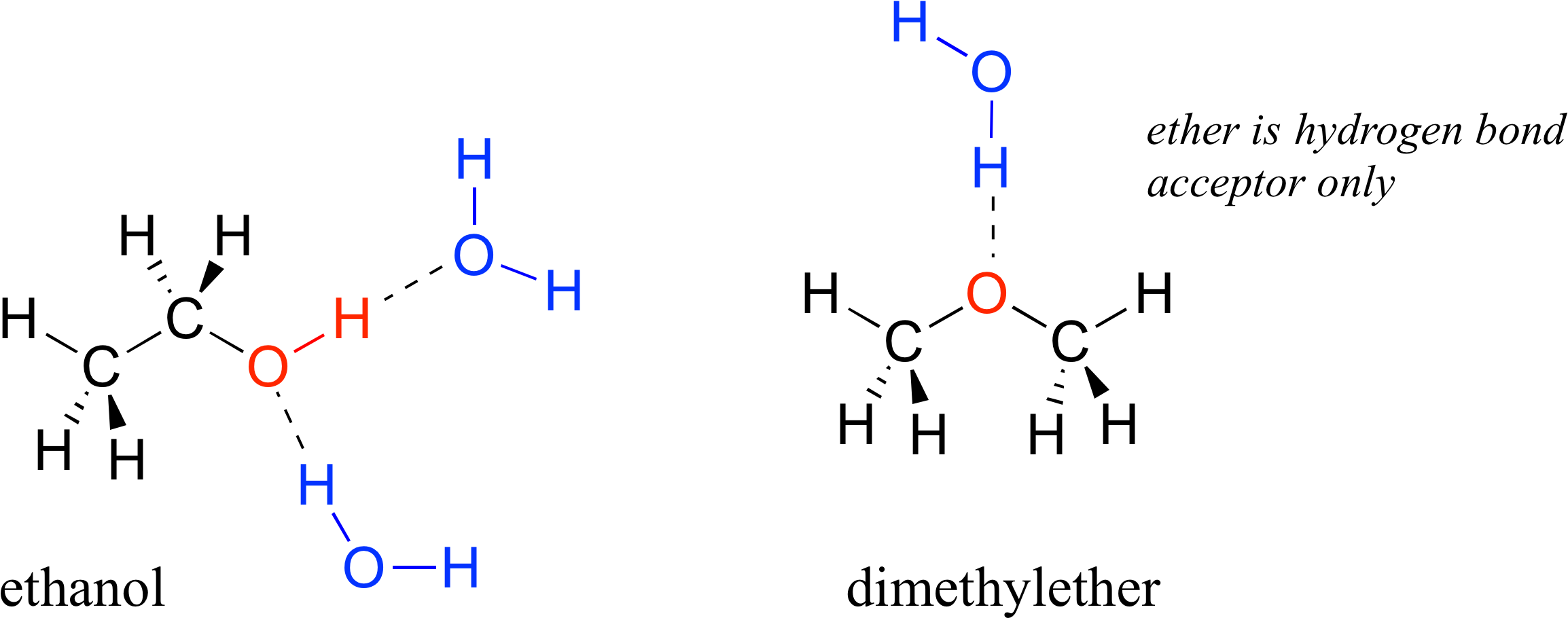
How many hydrogen bonds can an ethanol molecule form to neighbouring water molecules? Why? | Homework.Study.com

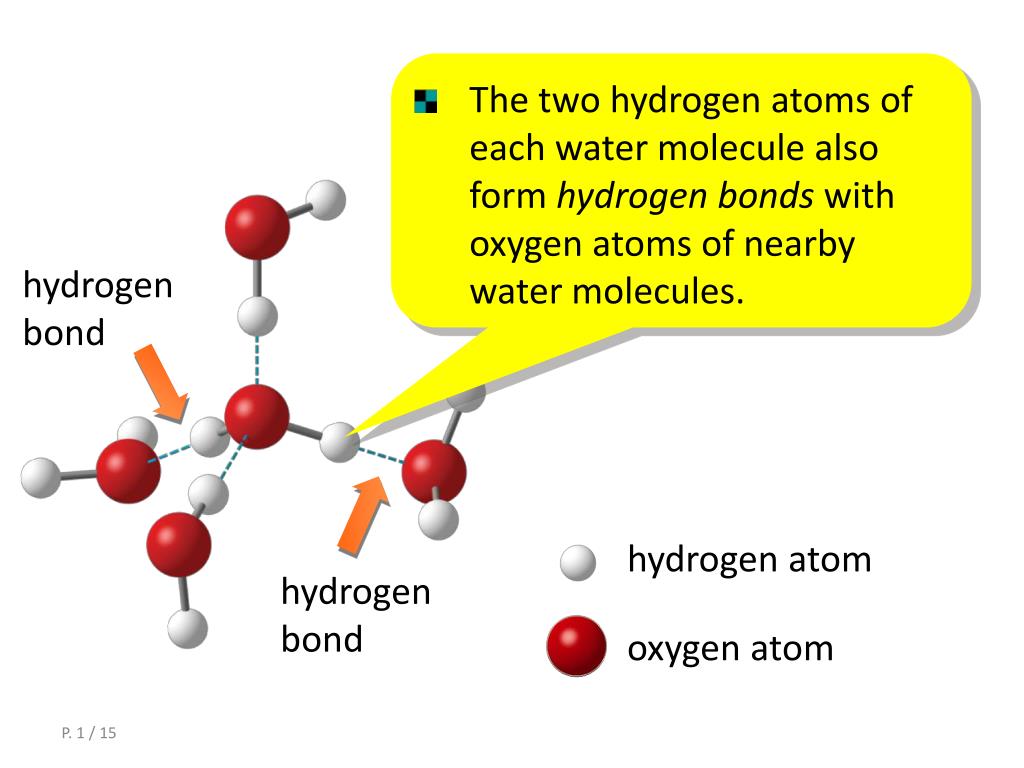
Think About Water 1.How many water molecules are present? (18) 2.How many Hydrogen bonds have formed between water molecules? (15) 3.Why can hydrogen bonds. - ppt download
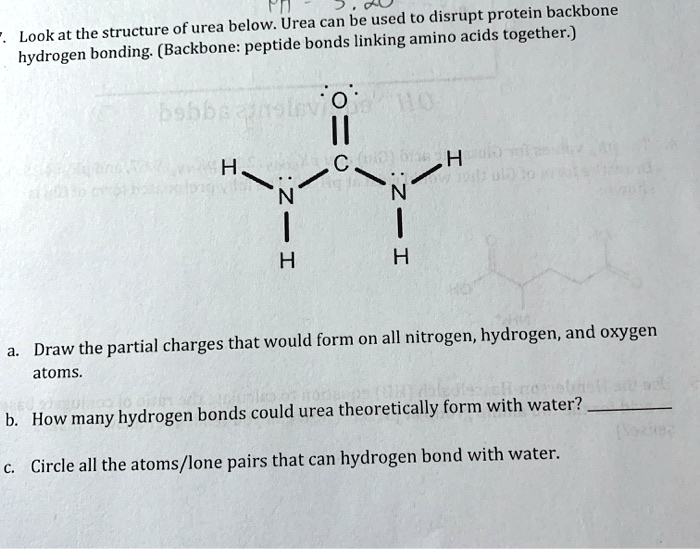
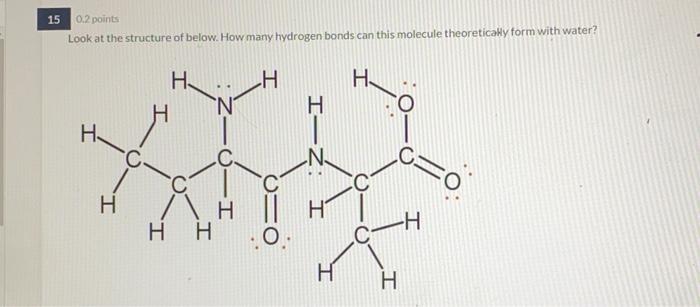
SOLVED: Fn be used to disrupt protein backbone Lookat the structure ofurea below: Urea can peptide bonds linking amino acids together:) hydrogen bonding (Backbone: H H Draw 'the partial charges that would

What is the maximum theoretical number of water molecules that one urea molecule can hydrogen bond with? | Homework.Study.com