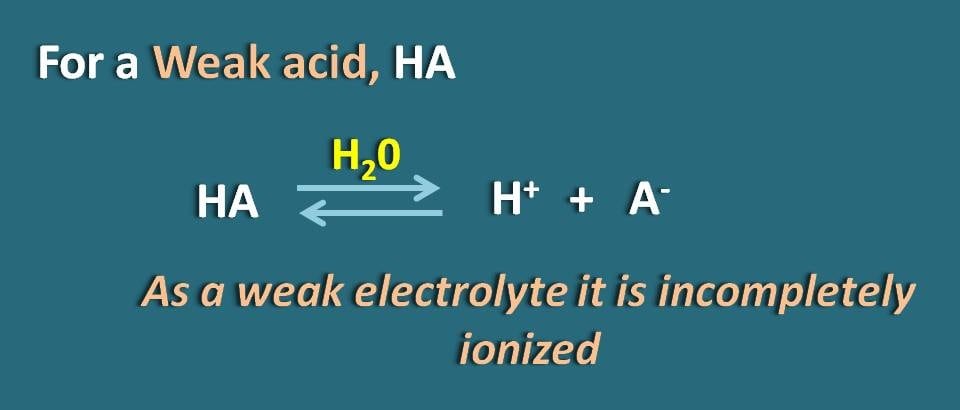
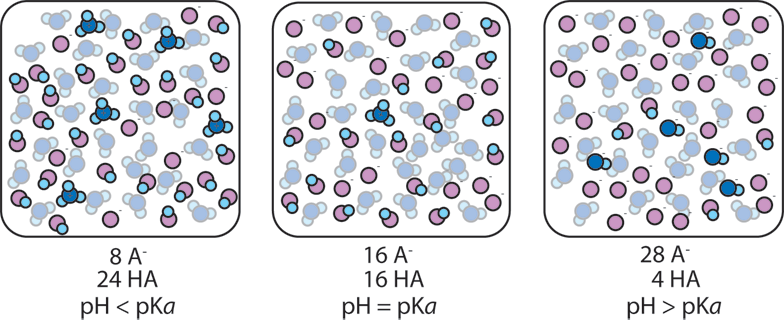
![SOLVED: What is the [A-] / [HA] ratio necessary to make a buffer solution with a pH of 4.14? Ka = 1.85 x 10-5 for HA. Show formula and calculation with units. SOLVED: What is the [A-] / [HA] ratio necessary to make a buffer solution with a pH of 4.14? Ka = 1.85 x 10-5 for HA. Show formula and calculation with units.](https://cdn.numerade.com/ask_previews/a61d22d0-b9a8-4b14-87ab-a744b7801b70_large.jpg)
SOLVED: What is the [A-] / [HA] ratio necessary to make a buffer solution with a pH of 4.14? Ka = 1.85 x 10-5 for HA. Show formula and calculation with units.

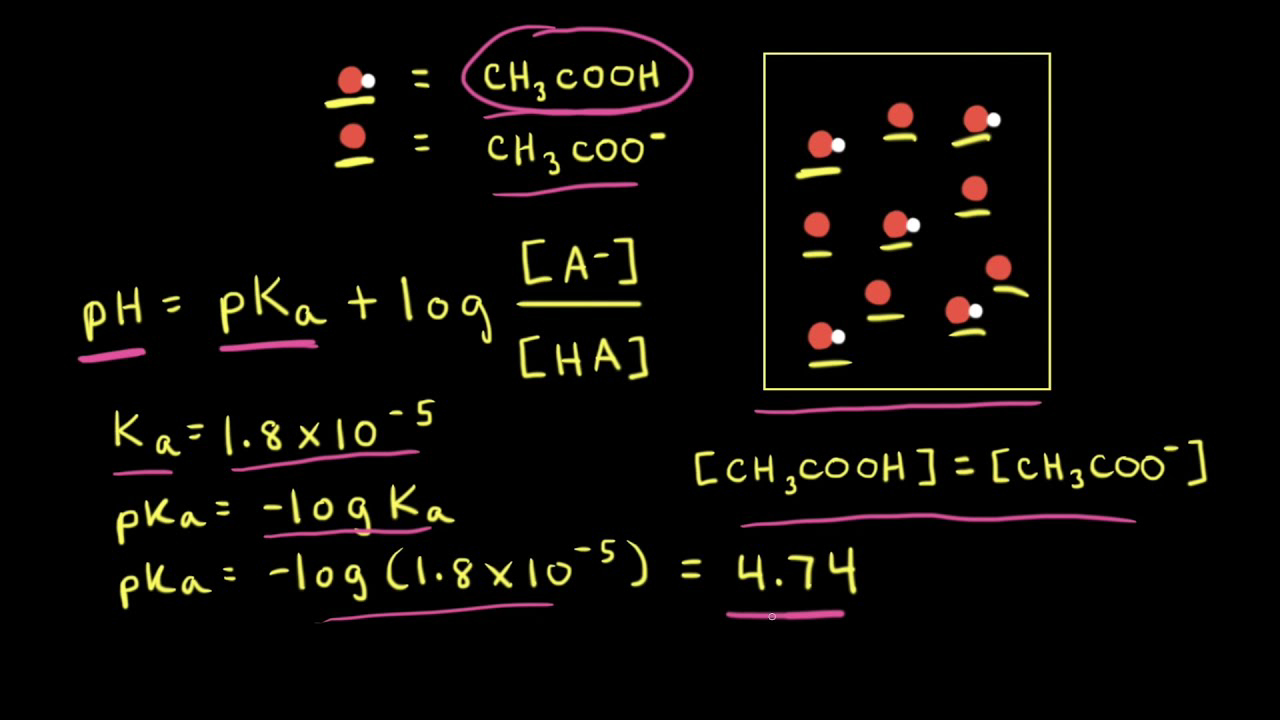
SOLVED: Use the Henderson-Hasselbalch equation to perform the following calculations. The Ka of acetic acid is 1.8 10-s Review your calculations with your instructor before preparing the buffer solutions Buffer A: calculate