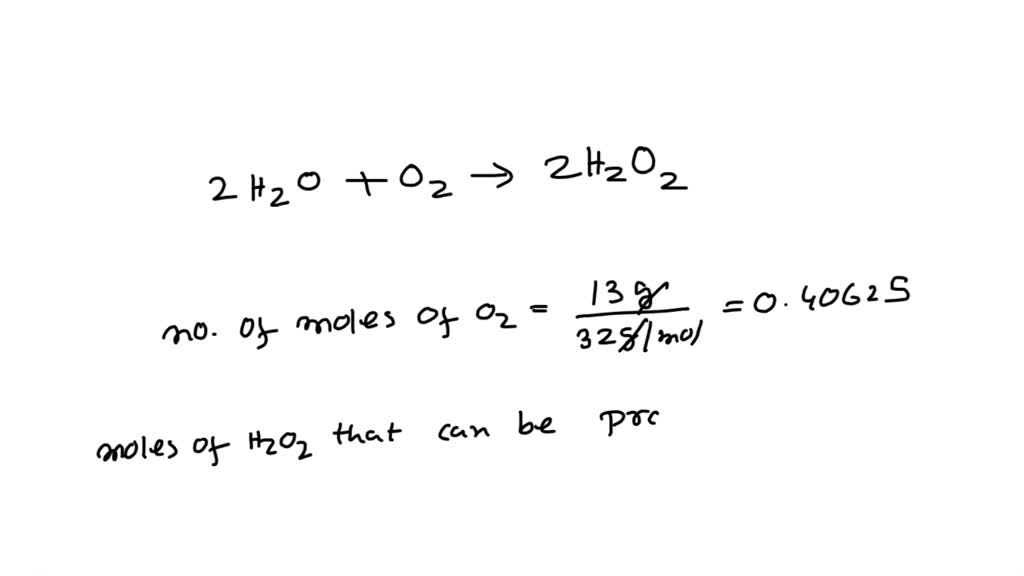
SOLVED: How many moles of H2O2 are produced in the production of hydrogen peroxide when 13 grams of oxygen are used?2 H2O+O2→2 H2O2 81 g H2O2 6.5 mol H2O2 0.81 mol H2O2 13 mol H2O2
In Situ OH Generation from O2− and H2O2 Plays a Critical Role in Plasma-Induced Cell Death | PLOS ONE

SOLVED: Balance the following equation: H2O2 HzO + 02 H202 HzO + 02 2 H2O2 2 HzO + 02 2 HzO2 2 H2O + 302 H2Oz 2 HzO + 02

Superoxide (O2⁻), hydrogen peroxide (H2O2), malondialdehyde (MDA) and... | Download Scientific Diagram















