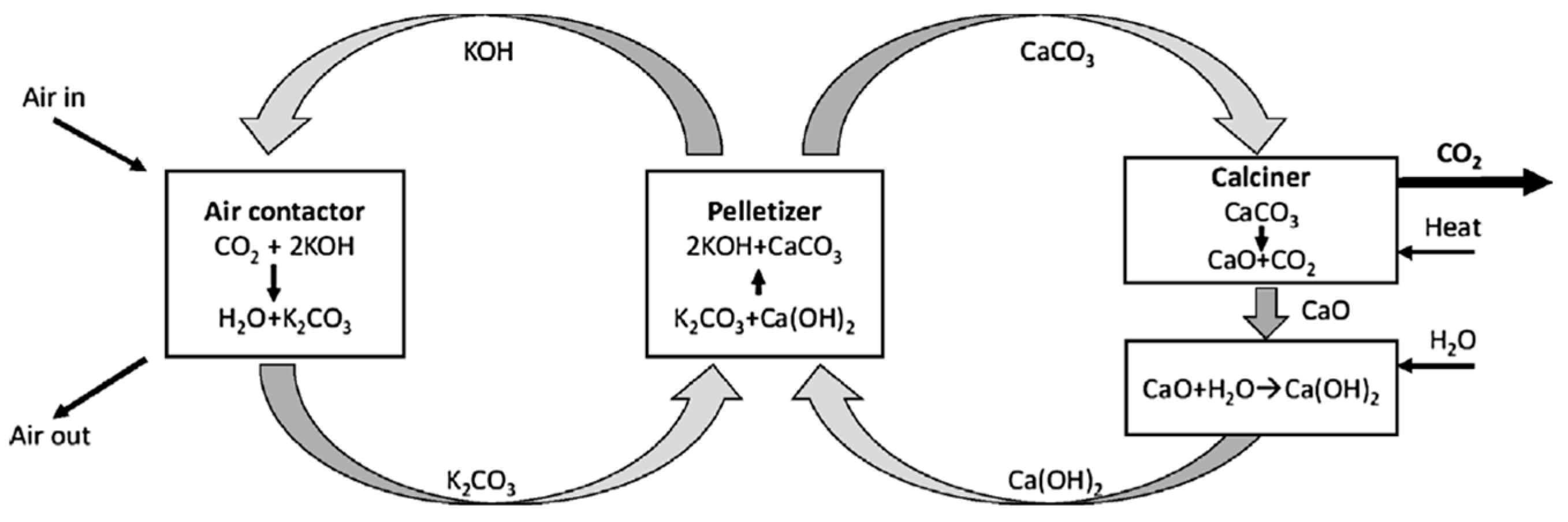
Crystals | Free Full-Text | Crystallization in Fluidized Bed Reactors: From Fundamental Knowledge to Full-Scale Applications

Electrochemical Splitting of Calcium Carbonate to Increase Solution Alkalinity: Implications for Mitigation of Carbon Dioxide and Ocean Acidity | Environmental Science & Technology
SOLVED: 00 M sodium carbonate IS needed lo prepare 250 0 mL ofa 0 125 M solution? What volume of 4 Attach File Browse My Computer @uesTion 10 Ifa 10.00 mL solution
SOLVED: You analyzed 2.475 g of a mixture of NaCl, SiO2, and CaCO3. You isolate 0.825 g NaCl, 0.825 g of CaCO3, and 0.580 g SiO2. You were told that the original

PDF) Harnessing Electrochemical pH Gradient for Direct Air Capture with Hydrogen and Oxygen By-Products in a Calcium-Based Loop

PDF) Electrochemical Splitting of Calcium Carbonate to Increase Solution Alkalinity: Implications for Mitigation of Carbon Dioxide and Ocean Acidity

Effect of Impurities on the Decarbonization of Calcium Carbonate Using Aqueous Sodium Hydroxide | ACS Sustainable Chemistry & Engineering
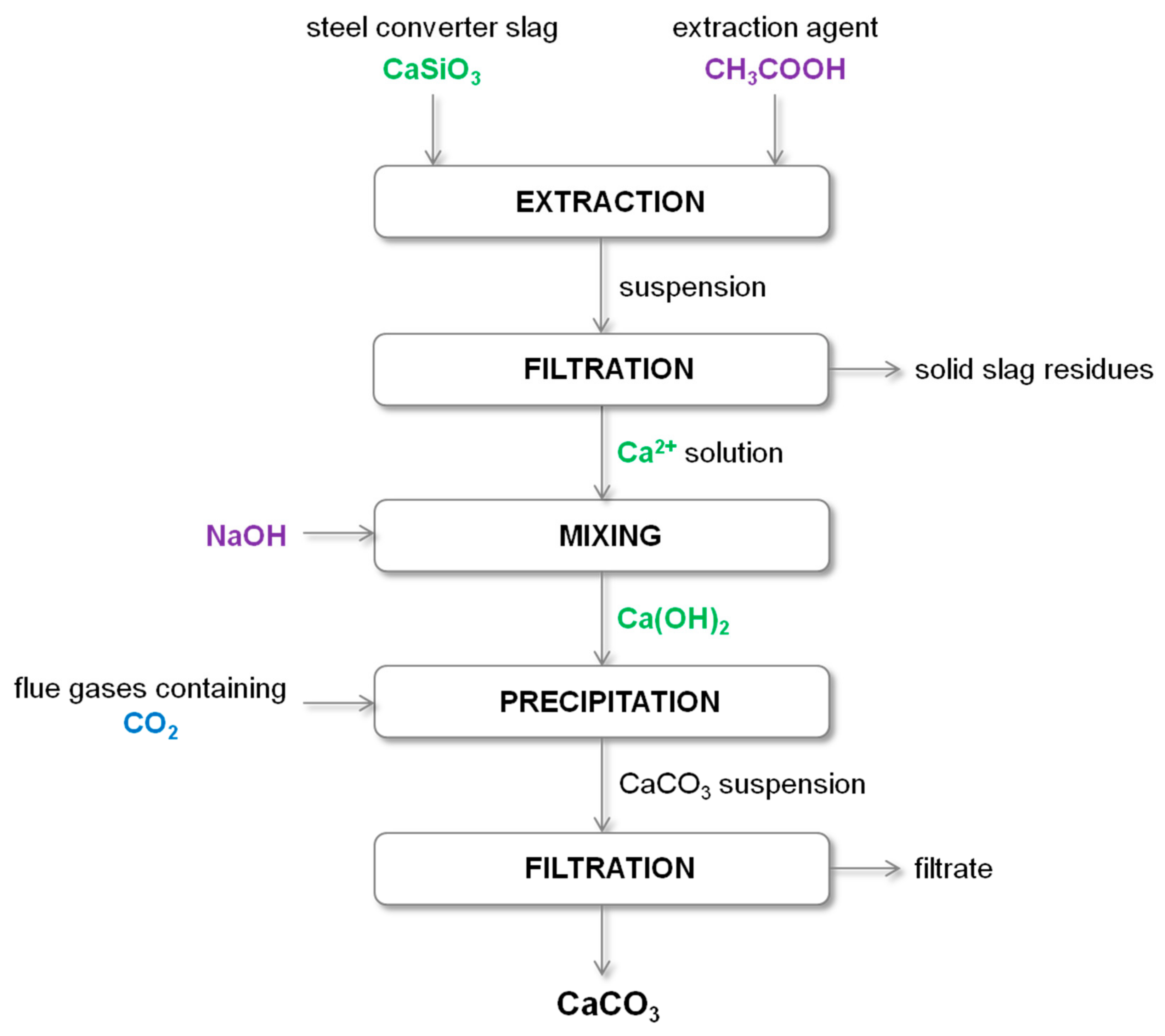
Energies | Free Full-Text | Utilization of Gaseous Carbon Dioxide and Industrial Ca-Rich Waste for Calcium Carbonate Precipitation: A Review

Integration of a calcium carbonate crystallization process and membrane contactor–based CO2 capture - ScienceDirect

Encapsulation of Proteins into CaCO3 by Phase Transition from Vaterite to Calcite | Crystal Growth & Design
Relating Structural and Microstructural Evolution to the Reactivity of Cellulose and Lignin during Alkaline Thermal Treatment wi

Integration of a calcium carbonate crystallization process and membrane contactor–based CO2 capture - ScienceDirect

Polymorph Selection and Structure Evolution of CaCO3 Mesocrystals under Control of Poly(sodium 4-styrenesulfonate): Synergetic Effect of Temperature and Mixed Solvent | Crystal Growth & Design
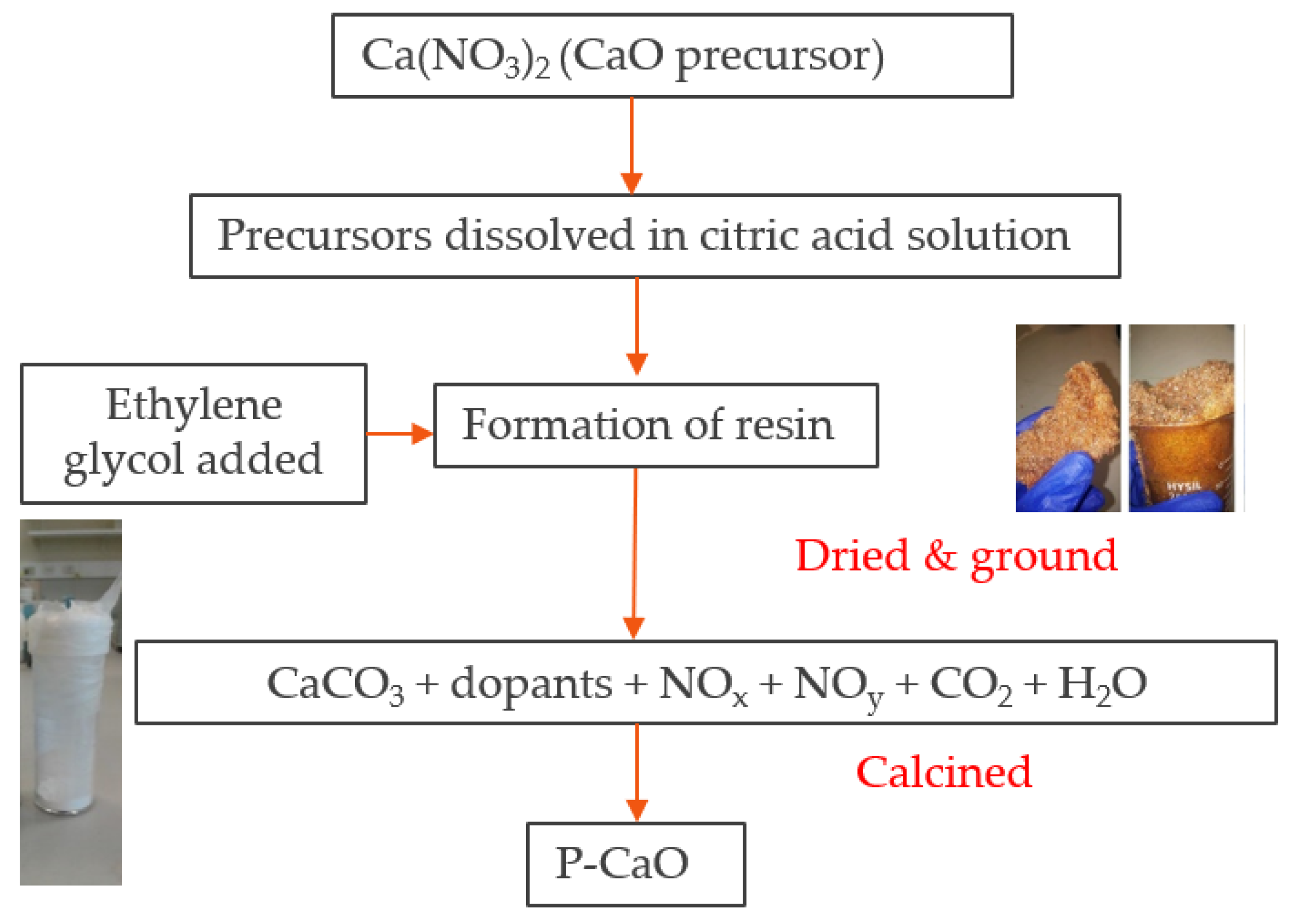
Applied Sciences | Free Full-Text | Comparative Kinetic Analysis of CaCO3/CaO Reaction System for Energy Storage and Carbon Capture










